A wide array of health problems are rooted in chronic inflammation. It’s a systemic condition that’s largely undetectable, unless you can convince your doctor you need a special blood test to look for it.
Inflammation is the process by which your body’s defence system – your immune system – protects the body from outside invaders such as bacteria, viruses or parasites. We are all familiar with an inflamed wound, for example, which is hot, red and painful.
Inflammation
But sometimes the immune system goes into overdrive and starts attacking our own body tissues, in the absence of invaders, producing inflammation in our joints, our muscles, even our brains.
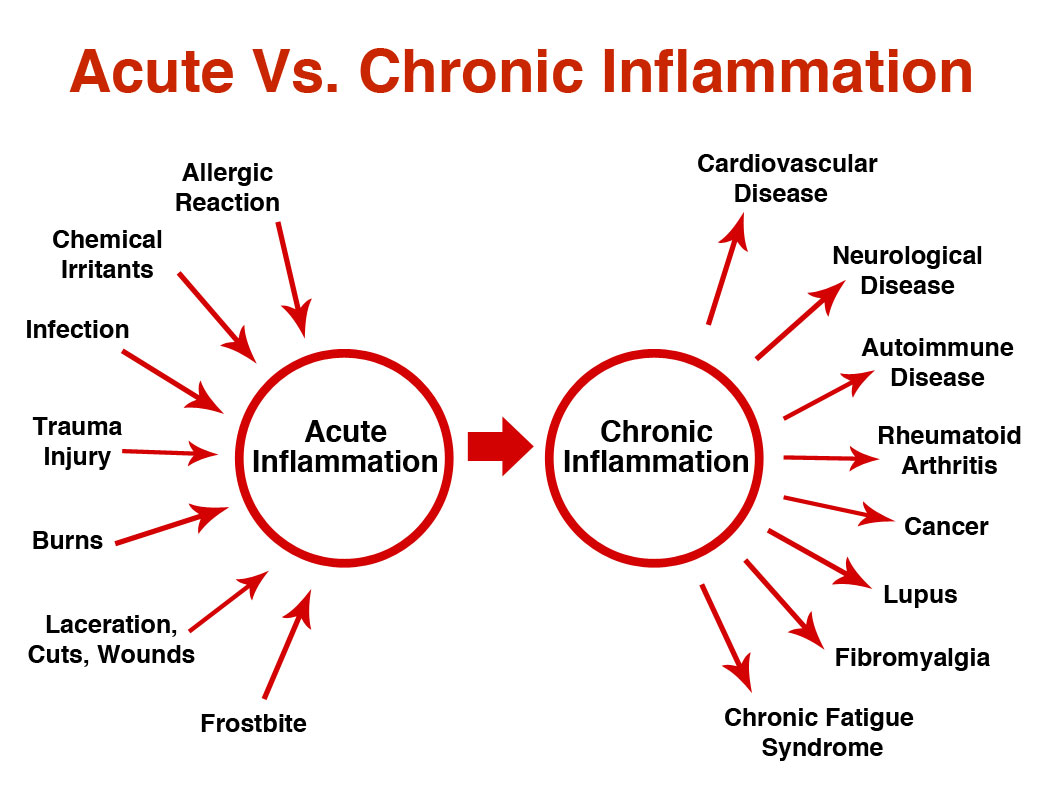
Inflammation can be acute (short-lived) or chronic (long term). In chronic inflammation the tissues remain inflamed for months or years, long after the initial trigger has gone.
Why does the body react in this way? We know that chronic inflammatory diseases are a relatively new phenomenon, and rising rapidly. Curiously it seems that the root of this dysfunction is in the immune system of the gut. And it is strongly linked to changes in the microbiome. (See disease begins in the gut)
Inflammation is the basic underlying process in all pathology, apart from deficiency diseases and injuries! That’s why the immune system/gut health is so foundational.
Many conditions
Chronic inflammation is linked to problems ranging from arthritis to Alzheimer’s, diabetes to hair loss.
Mary Harrington, PhD, director of the neuroscience program at Smith College, Massachusetts. One of the goals of her research is to find out why so many people are suffering from chronic fatigue. “We think that inflammation is key,” says Harrington.
Inflammation can also affect the central nervous system, which includes your brain and spinal cord.
- Addison’s disease
- ADD/ADHD
- Allergic reactions
- Alzheimer’s disease
- Ankylosing Spondylitis
- Antiphospholipid Antibody Syndrome
- Arthritis (osteoarthritis, rheumatoid arthritis (RA), psoriatic arthritis)
- Asthma
- Atherosclerosis
- Attention-Deficit/Hyperactivity Disorder
- Autism Spectrum Disorder
- Cancer
- Chronic Fatigue
- Chronic pain
- Coeliac disease
- Colitis (ulcerative)
- Crohn’s disease
- Dermatitis
- Diabetes
- Eczema
- Endometriosis
- Fatty liver disease
- Fibromyalgia
- Gastritis
- Gout
- Graves’ disease
- Gum disease
- Heart disease
- Hepatitis (active)
- Huntington’s disease
- Insomnia
- Inflammatory bowel disease (IBD)
- Interstitial cystitis
- Irritable bowel syndrome (IBS) – Crohn’s disease, ulcerative colitis, diverticulitis
- Lupus Erythematosus (systemic)
- Lymphedema
- Migraines
- Multiple Sclerosis
- Myalgic encephalomyelitis
- Myasthenia gravis
- Myositis
- Obesity
- Parkinson’s disease
- Peptic ulcer (chronic)
- Periodontitis
- Peripheral neuropathy
- Pernicious anemia
- Pleurisy
- Prostatitis
- Rheumatoid Arthritis
- Sarcoidosis
- Scleroderma
- Sinusitis
- Sjogren’s Syndrome
- Splenitis
- Stroke
- Systemic lupus erythematous (SLE)
- Thyroid issues
- Tuberculosis
- Vasculitis (autoimmune)
Inflammation and the brain
Chronic inflammation in the body can lead to inflammation in the brain. New research has led to important advances in our understanding of how inflammation can have important long-term implications for the brain, altering cognition, mood, and behaviour. The same processes that provoke the inflammatory response in the body also initiate the communication process to the central nervous system.
They accumulate in the bloodstream and thereby travel to the brain, where they cross into those regions in the brain where the barrier is weak. In addition, there are neural as well as blood-borne communication routes. For example, there are cytokine receptors on nerves, such as the vagus, that innervate peripheral immune organs, and these nerves, that communicate to the brain, are activated during infection.
Neuroinflammation
In other words, peripheral inflammation induces neuroinflammation – i.e. inflammation in the brain. During a normal infection, neuroinflammation and the resulting adaptive sickness behaviours persist only for a few days. However, if these responses become exaggerated or prolonged, the outcomes may well become established, leading to cognitive impairment instead of brief memory disruption, depression instead of reduced mood, chronic fatigue instead of inactivity, and chronic pain instead of acute pain.
Neuroinflammation is now a well-characterised feature of neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease and Huntington’s disease. Immune dysfunction outside of the central nervous system is also increasingly recognised as part of these diseases. Peripheral inflammation has emerged as a modulator of disease progression and neuropathy in general.
So what causes the inflammation in the first place?
The majority of inflammatory diseases start in the gut with a disordered microbiome – which progresses to systemic chronic inflammation. (See ‘Disease Begins in the Gut‘ and the following pages.) If the root of this immune dysfunction is a disordered gut microbiome, then the solution lies in seeking to repair or restore the microbiome.
